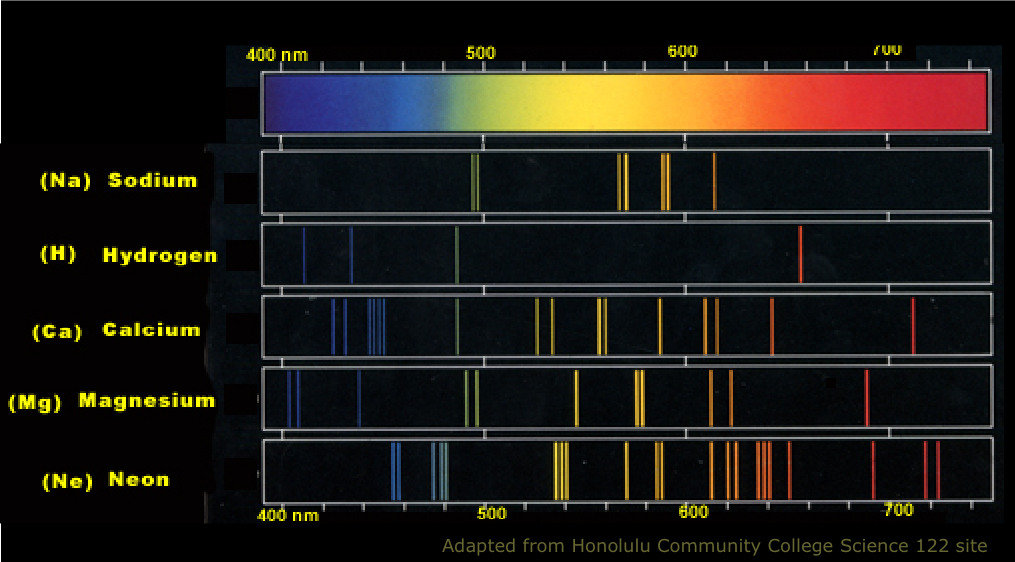
The Atomic Spectrum should be an absorption line Spectrum. The Atomic Spectrum should be the emission band Spectrum. The Atomic Spectrum should be a pure line Spectrum. The characteristics of the Atomic Spectrum are observed as:
#Atomic emission spectra series#
Spectral series are crucial in Astronomical Spectroscopy. The visible spectral lines in the hydrogen emission Spectrum are caused by Atomic transitions between distinct Energy levels. The Rydberg formula clearly splits the Atomic Hydrogen emission Spectrum into a number of wavelength-dependent spectral lines. An Electron emits or absorbs light of a specific wavelength as it jumps from one Energy level to the next.
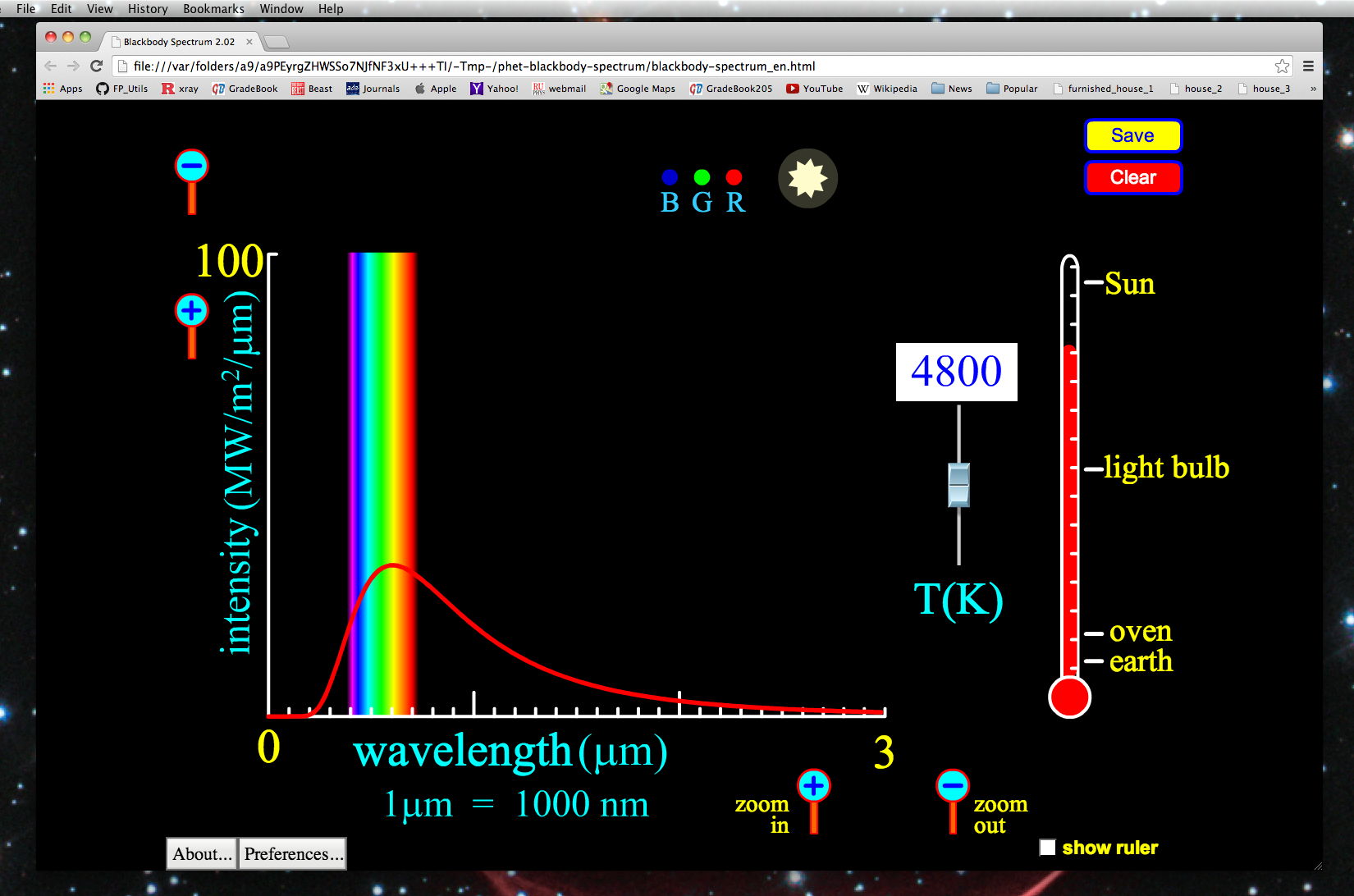
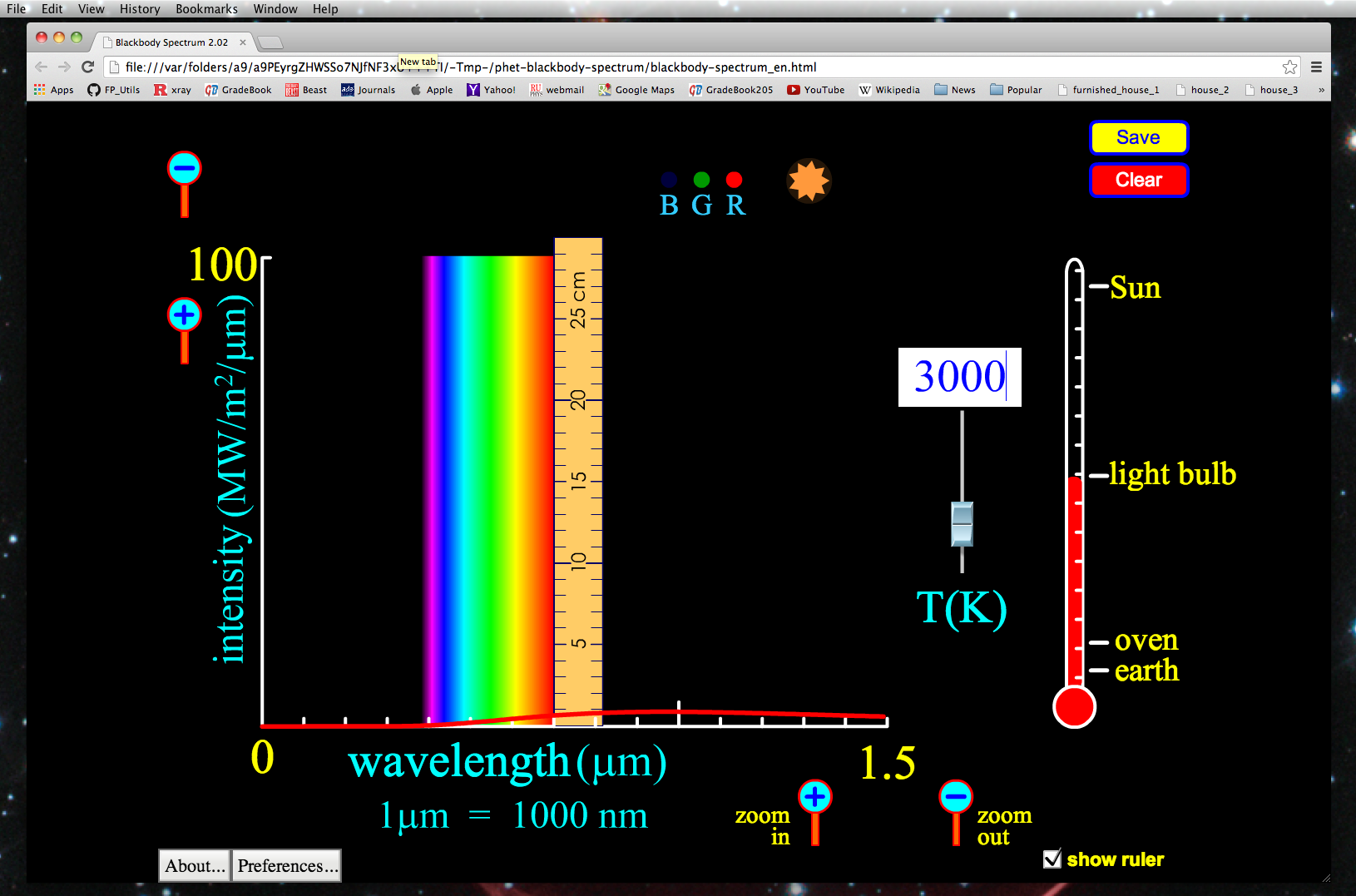
When an Electron gets excited from one Energy level to another, it emits or absorbs light of a specific wavelength.Īs an Electron moves between different Energy levels within an Atom, its Spectrum of Electromagnetic radiation is released or absorbed. When an Electron transitions from one Energy level to another, it emits light or photon with a specific wavelength. There are more Energy states in a tom than there are Electrons. In an Atom, Electrons have discrete and some specific energies. Each element has a characteristic Spectrum through which it can easily be recognized. 5.5.6), National Institute of Standards and Technology, Gaithersburg, MD.The Atomic spectra are defined as the Spectrum of frequencies of electromagnetic radiation emitted or absorbed during transitions of Electrons between Energy levels within an Atom. Data for the emission lines (wavelengths, transition strengths, energy levels, and statistical weights) are from Kramida, A., Ralchenko, Yu., Reader, J., and NIST ASD Team (2018).

"Polynomial Partition Function Approximations of 344 Atomic and Molecular Species" The Astrophysical Journal Supplement Series, 1981, 45, 621-633. "The Electronic Partition Functions of Atoms and Ions Between 7000 K and 12000 K" Spectrochimca Acta 1987, 42B, 1105-1111 and (c) For the Irwin model, Irwin, A. "The Electronic Partition Functions of Atoms and Ions Between 1500 K and 7000 K" Spectrochemica Acta, 1968, 23B, 521-525 (b) For the Tamaki model Tamaki, S. The calculated intensity for each of the element's emission lines from the NIST databaseĬoefficients for the partition coefficient models are from the following sources: (a) For the deGalan model, deGalan, L. The wavelength, in nm, for each of the element's emission lines from the NIST database The total line width of an emission line, in nm The model used to estimate the element's partition function The value of the partition function calculate for the specified temperature using the specified model Returns a list with the following components elementĪ vector of wavelengths, in nm, for which emission intensities are calculatedĪ vector of calculated absolute emission intensitiesĪ vector of calculated relative emission intensities See the package's vignette for additional details. The Irwin model is used in place of the Tamaki model for those elements (Be, B, Sc, and Ge) for which Tamaki does not report data. The simulation also requires a value for the element's temperature-dependent partition function, which is estimated using one of three models: the Irwin model, for temperatures between 1000 K and 1500 K, the deGalan model for temperatures between 1500 K and 7000 K, and the Tamaki model for temperatures between 7000 K and 12000 K. The simulation of an element's atomic emission spectrum relies on published characteristic values for each of the element's atomic emission lines, which are available from NIST: the wavelength, in nm, the energy of the upper energy level, in eV, the statistical weight of the upper energy level, and the transition probability,in s^-1. The total line width of an emission line, in nm. The temperature must have a value between 1000 K and 12000 K. The element of interest, entered using one of three options: the element's atomic symbol ( atom = "H"), the element's name ( atom = "hydrogen"), or the element's atomic number ( atom = 1). SimulateAES ( atom = "H", temperature = 7000, width = 0.3 )
